- Blog
- Elevate kingdom tales images
- 935 optical isolator
- Jihosoft file recovery 6-4 key
- Davinci resolve lite help
- Margarita recipes
- Blossom blast saga for pc
- Betrayal at krondor free download
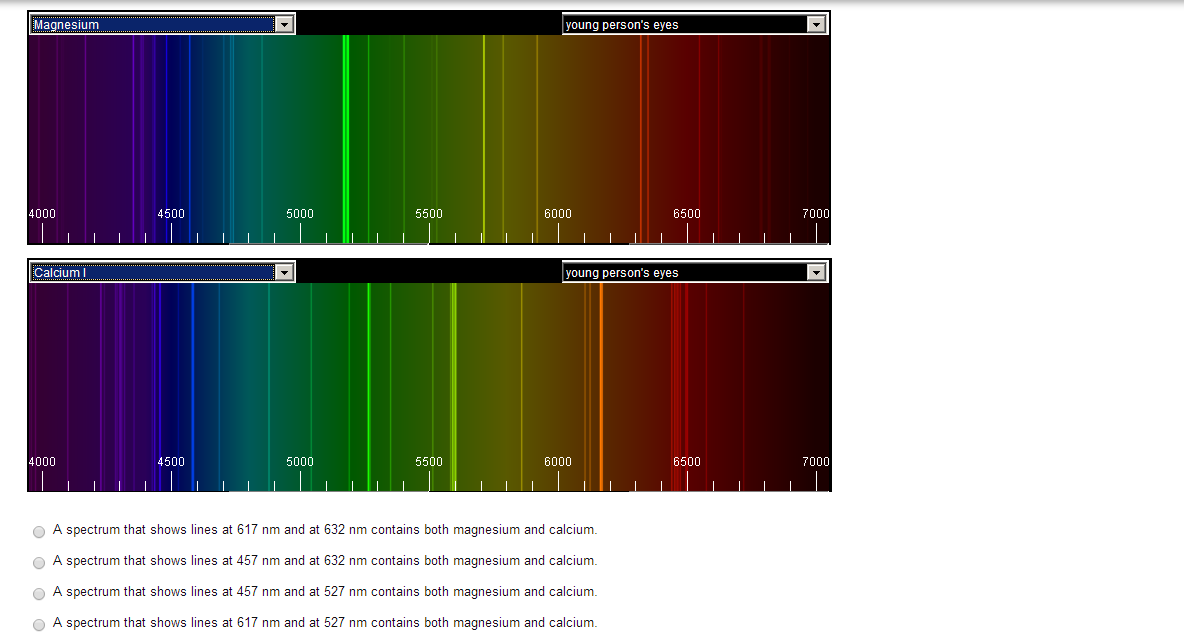
- Emission spectra
- Corel painter free
- Grid iron wars al wilson
- Macbook map music keys to google play music
- Popcorn time ios 14
Mechanism of fluorescence of 2-p-toluidinylnaphthalene-6-sulfonate, a hydrophobic probe, Biochemistry 5: 1908–1919.īrand, L., Seliskar, C. M., 1954, Fluorescent probes for conformational states of proteins.
:max_bytes(150000):strip_icc()/GettyImages-1096547948-35b3799817ca4b2fa06888893ef4a348.jpg)
Slavik, J., 1982, Anilinonaphthalene sulfonate as a probe of membrane composition and function, Biochim. R., 1954, Fluorescent indicators of absorption in aqueous solution and on the solid phase, Biochem. G., 1962, Universal intermolecular interactions and their effect on the position of the electronic spectra of molecules in two-component solutions, Opt. Dependence on the magnitude of the Stokes shift in the solvent luminescence spectrum (liquid solutions), Opt. G., 1962, Universal molecular interactions and theireffects on the position of electronic spectra of molecules in two-component solutions. G., 1961, Universal molecular interactions and their effect on the position of the electronic spectra of molecules in two component solutions I. Suppan, P., 1990, Solvatochromic shifts: The influence of the medium on the energy of electronic states, J. II, Effects of solvent medium on the absorption and fluorescence spectra, J. J., and Brand, L., 1971, Electronic spectra of 2-aminonaphthalene-6-sulfonate and related molecules. Kawski, A., 1992, Solvent-shift effect of electronic spectra and excited state dipole moments, in Progress in Photochemistry and Photophysics, J. A new method for the determination of dipole moments of molecules in fluorescent states, J. 61: 562–572.īaumann, W., and Bischof, H., 1982, Integral electro optical emission measurements. G., 1956, Theory of solvent effects of molecular electronic spectra. G., 1954, Solvent effects in organic spectra: Dipole forces and the Franck–Condon principle, J. S., 1950, The effect of the electrostatic polarization of the solvent on electronic absorption spectra in solution, J. Rettig, W., 1986, Charge separation in excited states of decoupled systems-TICT compounds and implications regarding the development of new laser dyes and the primary processes of vision and photosynthesis, Angew. Mataga, N., Kaifu, Y., and Koizumi, M., 1956, Solvent effects upon fluorescence spectra and the dipole moments of excited molecules, Bull. Von Lippert, E., 1957, Spektroskopische bistimmung des dipolmomentes aromatischer verbindungen im ersten angeregten singulettzustand, Z. J., 1989, Trans-4-dimethylamino-4-(1-oxobutyl)stilbene: A new fluorescent probe of the bilayer lipid membrane, J. Safarzadeh-Amiri, A., Thompson, M., and Krull, U.

This process is experimental and the keywords may be updated as the learning algorithm improves. These keywords were added by machine and not by the authors. Additionally, fluorophores often display spectral shifts when they bind to membranes. Alternatively, the ligand may induce a spectral shift in the intrinsic or extrinsic protein fluorescence. Binding is usually accompanied by a spectral shift due to the different environment for the bound ligand. Suppose a fluorescent ligand binds to a protein. However, there are many additional instances where solvent effects are used. This is accomplished by comparison of the emission spectra and/or quantum yields of the fluorophore when it is bound to the macromolecule and when it is dissolved in solvents of different polarity. One common use of solvent effects is to determine the polarity of the probe binding site on the macromolecule. Emission spectra are easily measured, and as a result, there are numerous publications on emission spectra of fluorophores in different solvents and when bound to proteins, membranes, and nucleic acids. These effects are the origin of the Stokes’ shift, which is one of the earliest observations in fluorescence. Solvent polarity and the local environment have profound effects on the emission spectra of polar fluorophores.
